Electrolytes under investigation
- Asker Jeukendrup
- Jul 6, 2022
- 4 min read
Updated: Mar 13
There are many discussions in sport, and people disagree on many topics, but one thing everyone seems to accept as common knowledge is that electrolytes are important. At the same time, it is also seems to be common knowledge that too much salt (or sodium) is not good for you… But aren’t electrolytes and salt just different names for the same thing?
We have probably all read how, if you sweat heavily and you see white salty marks on your clothing or skin, you are a “salty sweater” - a person who loses a lot of salt during exercise. Those individuals especially are often encouraged to take salt tablets, or at least an electrolyte-replacement drink. Electrolytes are recommended left, right and centre, and they’re a big business: you can get electrolyte drinks, dissolvable tablets, capsules or even chewing gum. In addition, electrolytes are sprinkled into many other products like energy gels and bars.
Do electrolytes improve performance?
Athletes worry they do not consume enough electrolytes during exercise. But let’s first go back to the question of WHY we would need electrolytes in the first place? Or even the question: do we need electrolytes at all? Many athletes have this belief that electrolytes are supposed to help performance, but how many studies do we have that show that this is the case? You will be shocked by the answer!
What is the evidence that electrolytes work?
I asked several people including Professor Ron Maughan and other leading researchers who spend a lot of their impressive careers researching sodium, but they couldn’t point me in the direction of articles that provided convincing evidence. So, I decided to start a series of blogs to address these questions and I have asked Dr Alan McCubbin to help, as he is one of the few researchers who has done extensive work in this area recently.
In the following series of blog posts, we will have a look at common beliefs and what the actual evidence is. In this first blog we will start with discussing what salt is, what electrolytes are and what is the role of sodium and some other electrolytes.
What is salt?
For most people salt refers to the white substance that we also know as table salt. Table salt is sodium chloride. A sodium ion (an ion is any atom or group of atoms that bears one or more positive or negative electrical charges) combines easily with a chloride ion. Sodium is positively charged (cation), chloride is negative charged (anion) so they attract and bind easily. There is one sodium cation (Na+) for every chloride anion (Cl–), so the chemical formula is NaCl. We often refer to salt as sodium, but sodium is just one part of a salt (40% of the weight of sodium chloride for example) and there are also salts that do not contain sodium at all (for example potassium chloride).

What are electrolytes?
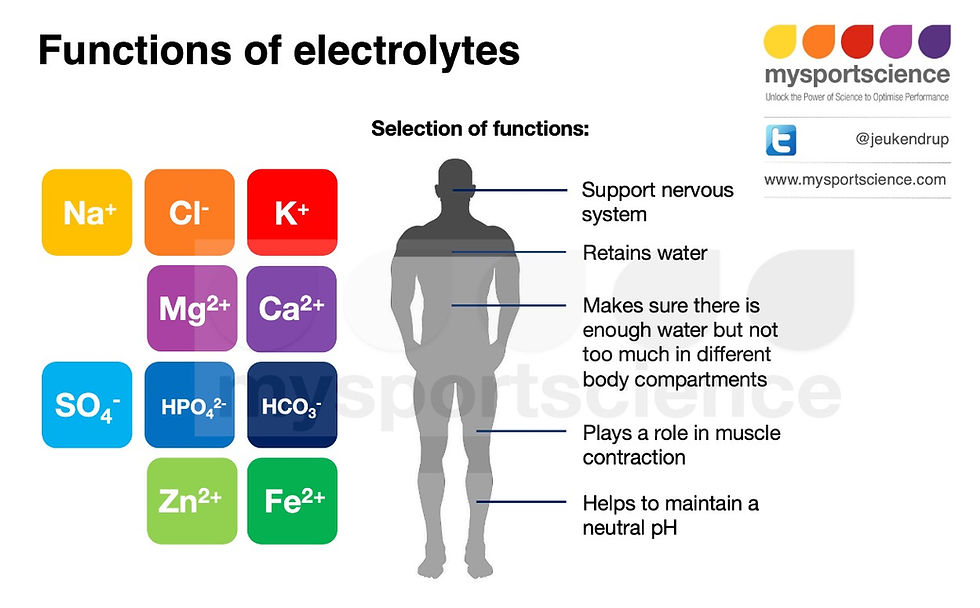
Minerals like sodium, calcium, and potassium become electrolytes when dissolved in water, meaning they can conduct an electric current in the water. A bit of chemistry: electrolytes, which include acids, bases, and salts, usually dissociate into their individual ions when dissolved in water, carrying either a positive charge (cation) or a negative charge (anion). Table salt (sodium chloride) for example dissolves in water and splits into individual sodium (cation) and chloride (anion) ions, and are therefore electrolytes. Each of the electrolytes are vital to many key functions in the body
Electrolytes are essential minerals like sodium, calcium, and potassium—that become electrolytes when dissolved in water, meaning they can conduct an electric current when dissolved in water.
The major electrolytes in the body fluids are sodium, potassium, chloride, bicarbonate, phosphate, sulphate, magnesium, and calcium. Of these, sodium, potassium, and chloride are found in the highest concentrations, although their distribution differs between the intracellular (within cells) and extracellular fluids (outside of cells, for example in blood). Sodium and chloride are found in higher concentrations in the extracellular fluid, whereas potassium is found in higher concentrations inside cells.
What is the role of electrolytes?
Electrolytes are important to the normal function of our bodies. Minerals not only form the building blocks of some tissues (for example calcium in bone) but are linked to a variety of essential functions: including oxygen transport, various enzymatic reactions and immune function. At least 20 mineral elements are known to be essential for humans, and 14 trace elements have been identified as essential for maintenance of health.
Essential in this context means that we need to obtain these mineral elements via our nutrition because they play an important role in our body’s functioning and we can’t make them in the body. Trace amounts of arsenic, nickel, silicon, tin, and vanadium may also be essential, but deficiencies or excesses (because of dietary sources) for these micronutrients are extremely rare.
Deficiencies of one or more of the minerals and trace elements result in symptoms of disease, and many deficiencies are also associated with immune dysfunction and increased incidence of infection. This is something that would develop if there was a consistent chronic shortage in the diet.
Electrolytes are often talked about in association with dehydration and mentioned in ads for sports drinks that promise to replace electrolytes lost through sweat. In the context of exercise performance, the main electrolytes of interest are sodium, potassium, and chloride because these are found in the highest concentrations in the body and also in sweat. Of these three, sodium is the most important one.
In the context of exercise performance, the main electrolytes of interest are sodium, potassium, and chloride because these are found in the highest concentrations in the body and also in sweat.
In the next few blogs we will discuss: