The role of sodium during exercise
- Asker Jeukendrup
- Jul 7, 2022
- 4 min read
Updated: Mar 13
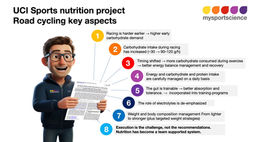
Sodium is said to be important for athletes, and different arguments are used to explain why it is so essential. We will dive into the evidence a little more but the infographic below will already provide a short summary of the analysis.

Sodium plays several essential roles during exercise. Firstly, sodium plays a crucial role in water balance. It does this due to its effect on osmolality of the extracellular fluid (fluid in the bloodstream and surrounding the outside of cells). Osmolality is the number of dissolvable particles within the liquid that it’s dissolved in. In the body, sodium is the greatest contributor to the number of particles in the extracellular fluid. A drop in the sodium concentration in the blood (hyponatremia) means a drop in osmolality, and this can have serious consequences for fluid balance. If the concentration of sodium in the blood drops, fluids will move into tissues to even out the osmolality between the inside and outside of cells. One of those tissues is the brain and this can result in brain swelling and even death, but this requires both reduced osmolality to direct water into the cells, as well as excessive total water to cause the cells to expand to dangerous levels. So whilst this is thankfully rare, clearly it is important to make sure that sodium concentration in the blood is well maintained.
Does sodium increase water absorption?
Sodium is often part of drinks that are designed for consumption during exercise (carbohydrate-electrolyte drinks or sports drinks). There are a number of reasons why carbohydrate and sodium are added to these drinks. Some glucose can increase the absorption of sodium, and water will follow the sodium and glucose as a result of osmotic drag. It is sometimes suggested or claimed that sodium helps water absorption, but this effect is only very small. To illustrate this I want to show you the data of one study we performed. In this study different amounts of sodium were provided in a 6% glucose solution.
The Na group ingested the following beverages:
Na0: 6% Glucose
Na20: 6% Glucose + 20 mmol/L sodium = 460mg/L (roughly 0.5 g/L)
Na40: 6% Glucose + 40 mmol/L sodium = 920 mg/L (roughly 1.0 g/L)
Na60: 6% Glucose + 60 mmol/L sodium = 1380 mg/L (roughly 1.5 g/L)
You can see the results in the figure below. If there are differences with moderate to very large amounts of sodium in a drink, the effects are minimal.

As can be seen from this figure there may be small differences between drinks (not significant in this study) and this sodium has no major effect on water absorption. But this doesn’t mean that sodium doesn’t have a role to play in hydration during exercise.
Sodium improves the taste of drinks and stimulates consumption
Sodium tends to improve the palatability of drinks. Sodium also maintains the thirst response, because thirst is triggered by increased osmolality in the blood, and so stimulates drinking in that way. This means that athletes are more likely to drink and this is of course a prerequisite to staying hydrated. It is important to note that in order to make a drink more palatable, we only need small amounts of electrolytes. 400-500 mg sodium per liter will make a drink more palatable, but more than 1000 mg per liter will make it less palatable.
Sodium increases beverage osmolality
One thing I would like to point out here is the role of electrolytes on the osmolality of fluids that we drink. Although the effects of osmolality of drinks and ultimately stomach content may be overrated in many conditions as well, many believers in electrolytes are also believers in an important role for osmolality. Those individuals often spend a fortune on isotonic drinks (those that have a similar osmolality to the blood) and then they supplement with salt tablets or electrolyte tabs. These tabs will take the osmolality through the roof and any small potential effect of isotonic drinks will be gone…
Pre-exercise sodium and water retention
Finally, sodium is instrumental in the retention of the ingested water in the body’s water pools. So, essentially, sodium will help to store water in the body. Although this is a theoretical benefit, there is little evidence that this can actually help performance in most situations. The only evidence comes from studies in extremely hot and humid conditions (often with minimal wind cooling). In these studies, sodium and other solutes such as glycerol were used to preload water to improve temperature regulation during exercise. This is a different scenario than drinking a solution with electrolytes during exercise though. The studies that use pre-loading, typically start 2h before exercise with a large dose of salt or glycerol and a very large volume of water (2-3L).
Theories why sodium is important
The two main theories and assumptions on which the case for sodium supplementation is built is that you lose a lot of sodium during exercise (this is an assumption in most cases) and therefore:
There might be an effect on blood sodium concentration or
Body sodium stores are affected so that other roles of sodium are compromised.
The infographic at the start of this blog lists a number of claims that are made about sodium and the actual evidence. Alan McCubbin will explore these claims in more detail in the next few blogs.
In summary, sodium does not help water absorption from drinks in any major way (glucose is much more important for this). However, sodium will make a drink taste better and make you thirstier. Therefore, you are more likely to drink and this will of course help you to stay hydrated (but you probably don’t need salt tablets for this and just adding small amounts of sodium to a drink may be more effective).
The more important effect of sodium is the role in maintaining fluid balance between various body compartments (in particular the intra- and extracellular space). Whether you need to take electrolytes during exercise to achieve this is a question we will address in more detail here.